Connect with a Global Community of 90+ Oligo Experts
As oligonucleotide therapeutics advance across ASOs, siRNA, gRNA, and increasingly complex conjugates, the industry faces mounting pressure to ensure robust analytical characterization, scalable manufacturing, and globally aligned CMC strategies. Biopharma leaders are actively seeking partners who can support with high-quality raw materials, advanced analytical technologies, synthesis and scale-up capabilities, impurity control, and regulatory-ready CMC frameworks that can withstand increasing global scrutiny.
This is why the 5th Oligonucleotide Analytical Development & CMC Summit stands as the industry’s only dedicated forum uniting analytical development, CMC, quality, regulatory, and process leaders across pharma, biotech, and academia.
The demand for innovative technologies and services that enable deeper molecular understanding, improve impurity identification, and strengthen control strategies across the development lifecycle has never been greater.

Partnering with us provides a powerful platform to:
- Position Your Organization at the Forefront of Analytical Innovation & CMC Excellence in Oligonucleotide Therapeutics
Gain visibility alongside leading biopharma advancing complex oligo modalities, by aligning your brand with cutting-edge discussions on impurity profiling, advanced analytics, and scalable control strategies - Showcase Your Solutions to Enable Robust Characterization, Control, & Regulatory Readiness
Demonstrate how your technologies and services support accurate impurity identification, analytical comparability, and phase-appropriate control strategies, critical to building successful regulatory submissions - Connect Directly with Senior Analytical, CMC, Quality, & Regulatory Leaders Actively Seeking Solutions
Engage with decision-makers across pharma and biotech who are tackling real-time challenges in oligo development, creating meaningful, high-value partnerships beyond surface-level interactions
By partnering with this summit, you will not only gain visibility among the most influential stakeholders in oligonucleotide development but also play a central role in shaping best practices for analytical development and CMC strategies that underpin successful, scalable, and regulatory-compliant oligonucleotide therapeutics.
What to Expect?
Raise Your Brand Awareness
Position your organization at the forefront of oligo AD & CMC innovation to ensure your brand is seen by senior decision-makers from global pharma, biotech, and regulatory bodies actively shaping the standards for oligo therapeutics.
Distinguish Yourself from the Crowd
Learn from our unrivalled market intelligence of where key pain points lie for potential clients and tailor your presentations in line with their needs. Demonstrate real-world value through thought-leadership sessions, case studies, and live discussions with key stakeholders.
Generate Commercial Opportunities
Engage directly with prospective partners, collaborators, and clients actively seeking solutions to their analytical and CMC challenges. From early discovery to GMP readiness, build new business relationships and accelerate your commercial pipeline through qualified, face-to-face connections.
Uncover the Next Market Trend
Gain firsthand insights into emerging analytical, manufacturing, process, quality control, and regulatory priorities, that are driving the next generation of oligos. Stay ahead of competitors with intelligence from global innovators and regulators defining the future of mRNA.
Brush Shoulders with Industry Experts
Network with leading scientists, directors, heads, CxOs, and regulatory authorities in an interactive, discussion-driven setting. Exchange ideas, benchmark strategies, and establish meaningful collaborations with the experts setting the agenda for Oligonucleotide Analytical Development and CMC worldwide.
Key Services & Solutions
Our expert attendees from oligo biopharma are looking for service and solution providers with capabilities in the below areas but not limited to:
CDMO & Manufacturing Enablement
End-to-end development and manufacturing capabilities that support oligonucleotide programs from early-stage process development through GMP production. From tech transfer and scale-up to clinical and commercial supply, enable robust, compliant manufacturing strategies that address synthesis complexity, improve scalability, and reduce risk across diverse oligo modalities including ASOs, siRNA, and conjugates.
Raw Materials & Synthesis Inputs
High-quality phosphoramidites, resins, reagents, and critical starting materials that underpin consistent and efficient oligonucleotide synthesis. Addressing challenges in supply reliability, purity, and batch-to-batch consistency, these solutions are essential to building strong upstream control strategies and ensuring downstream product quality and regulatory compliance.
Analytical Technologies & CRO Services
Advanced analytical instrumentation and outsourced expertise enabling precise characterization of oligonucleotide drug substances and products. From LC-MS and HILIC to NMR and orthogonal methods, these solutions support impurity identification, structural elucidation, and method development to meet increasing regulatory expectations and enable confident progression through clinical development.
Software, Data, & Analytical Intelligence
Digital platforms and data-driven tools designed to streamline analytical workflows, automate data interpretation, and enhance process understanding. By reducing manual data deconvolution and enabling faster, more accurate insights, these solutions support stronger control strategies, improved reproducibility, and more efficient decision-making across analytical development and CMC.
Hear What Our Past Sponsors Have to Say

The intimate group size fostered meaningful networking and made it easy to connect with fellow attendees. The sessions were thoughtfully curated, offering a diverse range of perspectives on the future of the field and the evolving regulatory landscape
Supriya Kadam, Associate Director ECO Process Development, Codexis

Discussing with experts in a small group really helped me expand my network.
Taisuke Ichimaru, Contract Development & Manufacturing Organization Business Manager, Ajinomoto Bio-Pharma Services
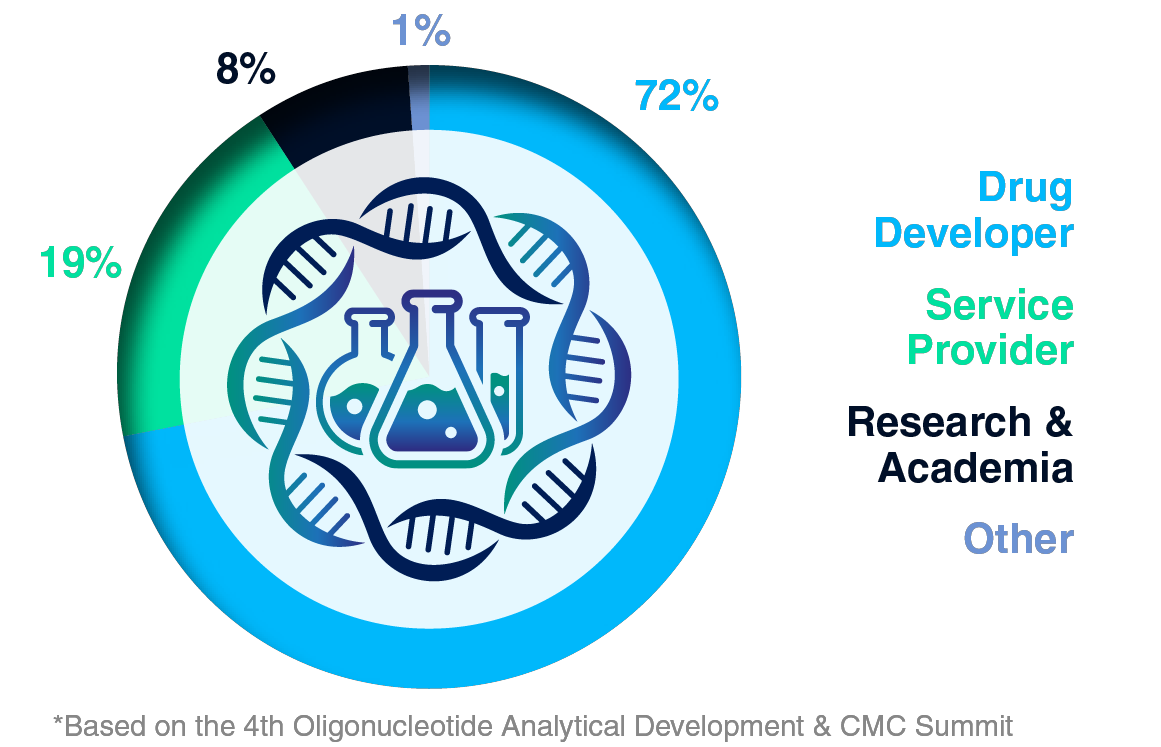
Audience Composition
Company Type
Attendee Seniority

Attending Companies Include









Get in Touch
Take advantage of our bespoke sponsorship opportunities to achieve your commercial goals. Email us if you would like to get involved and discuss a bespoke package suited to your needs.

Katie Orchard
Senior Partnerships Director